 When fully implemented, the label of most devices will include a unique device identifier (UDI) in human- and machine-readable form, which will ultimately improve patient safety, modernize device postmarket surveillance, and facilitate medical device innovation. The FDA established the unique device identification system to adequately identify medical devices sold in the United States from manufacturing through distribution to patient use. This new date provides a 75-day extension of an existing FDA compliance policy published in the July 2020 version of this guidance. I/LS/LS devices, including Class I I/LS/LS devices, are already expected to comply with GUDID submission requirements. Additionally, the FDA does not intend to enforce the GUDID submission requirements for Class I and unclassified devices, other than implantable, life-supporting or life-sustaining devices, regardless of whether they are consumer health products, before December 8, 2022. This final guidance describes the FDA's compliance policy regarding GUDID submission requirements for certain Class I devices considered consumer health products. The update to this guidance reflects the finalization of the draft guidance "Select Updates for Unique Device Identification: Policy Regarding Global Unique Device Identification Database Requirements for Certain Devices", which was issued October 14, 2021, and following the consideration of public comments. you get the point, too many words in parentheses and would be distracting, take a look.On July 22, 2022, the FDA posted the final guidance: Unique Device Identification: Policy Regarding Compliance Dates for Class I and Unclassified Devices, Direct Marking, and Global Unique Device Identification Database Requirements for Certain Devices. This final guidance describes the FDA's compliance policy regarding Global Unique Device Identification Database (GUDID) submission requirements for certain Class I devices considered consumer health products. So in other words, the ( ) are already everywhere, running throughout the entire script, if (CONT"D) is added on top of that. For those new to screenwriting, Final Draft is a word processor designed to format screenplays for movies and television. Just scroll though the script and you will see that Boal didn't skimp in using wrylys to his advantage in telling his story and structuring his script. Here are your character names, your locations. x is a term in Regular Expressions Character Classes for an hexadecimal digit. Within the Final Draft software program, every time you type in a name in the Character element or type a new Scene Heading, it’s added to your SmartType, the auto-fill function in Final Draft that remembers them for you. Although if you look at something like "The Hurt Locker (2009) great script, it seems Mark Boal was wise enough to keep (CONT"D) out of his script due to the fact that there would be a lot of Character ACTIONS coming through in wrylys. The Lists tab shows you the specific entries that Final Draft has memorized, organized by style element. Final Draft software trick 1: rebuild your smart type. page 2 and so on.or 2017 Horror remake in "It" uses CONT"D throughout and there is a lot of dialogue in that script between the child characters. 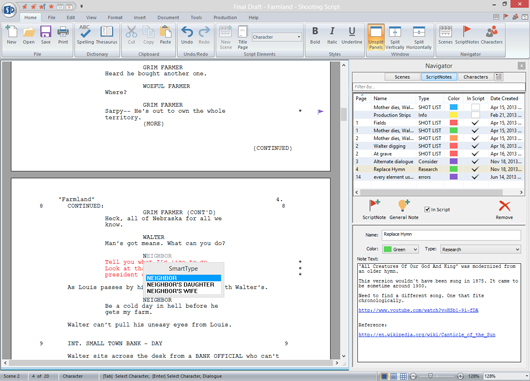
Use them as character inspiration, to start your own story, or borrow pieces to generate. For example in the script for "Thor: Ragnorak" (2017) you will find them on page 1!. Then, polish up your final draft on the next round of edits. If you scroll through most modern day scripts you'll find that most of them use (CONT"D) following a character's name during ongoing dialogue between action. Track data specific to each scene in this new floating palette such as the scenes story beats that will eventually make up the Action, Characters and Dialog of. 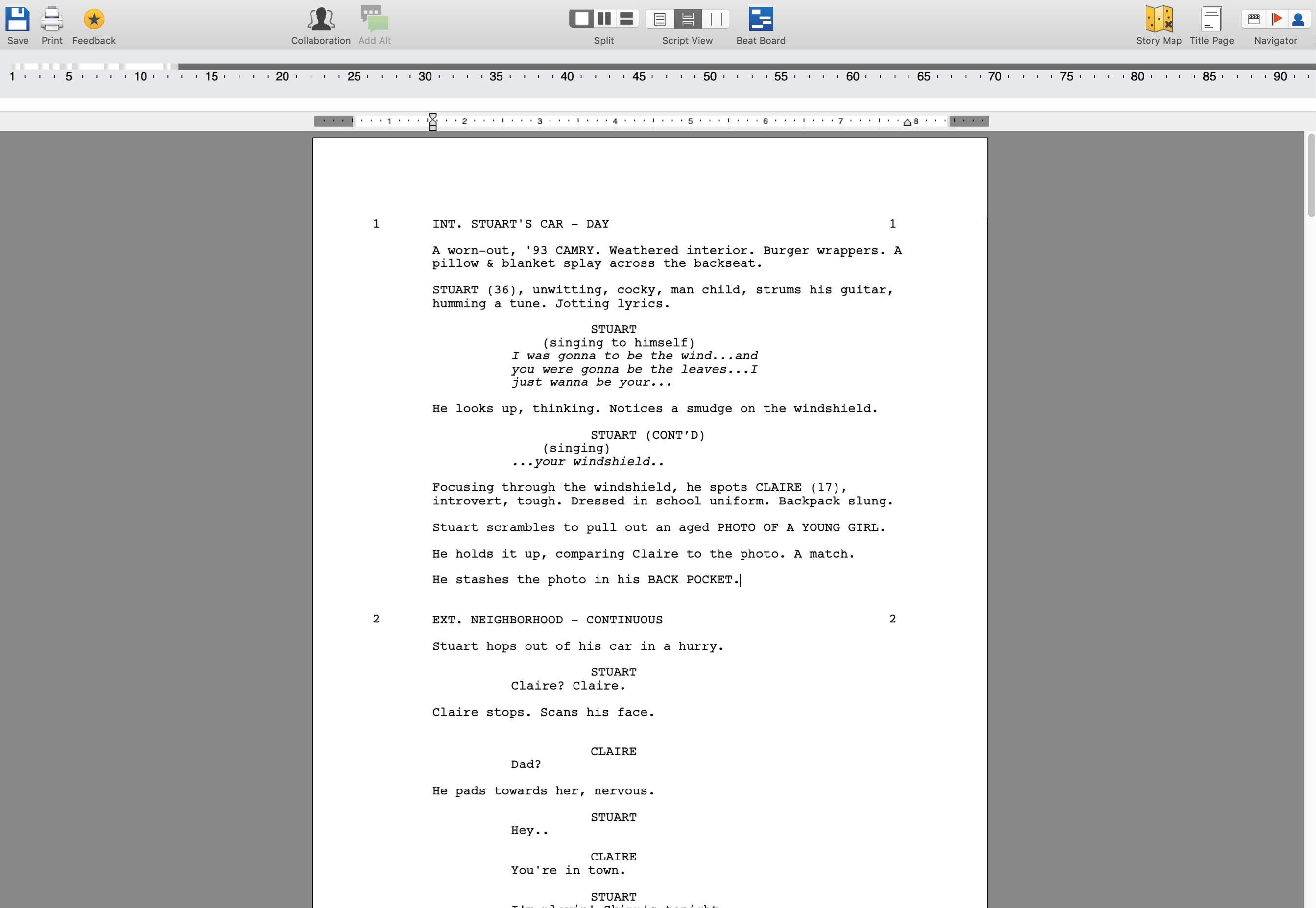
It seems the majority of writers keep them in. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
